Mercury and heavy metals discarded into the waste stream have a strong impact on our ways of live. Here, we consider how discarding fluorescent lights impacts environmental contamination.
This post is more heavily documented than our other posts because the results are likely to make true CFL believers angry. Please, evaluate and comment on the facts; TalkRadio-type flame comments will not make it to the comment section. Don’t forget, you must stand behind what you say, anonymous comments are not published, either.
Jump to Mercury Waste . Jump to Rare Earth Waste . Jump to Summary .
____________________________________________________
Mercury (Hg) Waste
Mercury will permanently disrupt the nervous system of those who ingest it or breath its vapor. Young children are susceptible to damage by very small quantities. A main way to enter the population is in the form of methylmercury, CH3Hg.
The US Geological Survey discusses why in many fish populations, and why eating a lot of certain types of fish is not in the best interests for most adults; pregnant women should avoid it. Many web sites offer additional information on mercury. It is interesting that Hg has a 70 day half life, that is 1/2 of the current amount in the body will be eliminated during the next 70 days, so contaminated fish can become clean, if kept in clean water.
Descriptions of how Hg enters the environment are enlightening. Elemental Hg evaporates easily at room temperature. About 6% of metallic Hg will vaporize immediately when a tube is broken. Hg also combines to form HgO which is absorbed by breathing. If a bulb breaks, open the window and leave the room. Once in aerosol form, Hg does not interact easily with other chemicals and it has a long dwell time in the atmosphere, allowing a single point emission site to affect the entire globe. This means that airborne Hg is the one of the most difficult pollutants to deal with.
Up to 40% of the will leach into the landfill within a few months, and a large fraction will be bound onto the tube’s inner wall. If shattered and the shards are in contact with the soil and groundwater, all the Hg must leach out into the environment, given a long enough time. If intact, it ought not spread from the sealed capsule. This sounds reasonable, but I have not located published data to document timelines. The USGS speculates that acidic pH helps Hg dissolve into water.
That was a long discussion to explain Hg worries → I assume that if x tons of Hg are put into the environment, all x ultimately endangers our children.
Sources of Hg
The table here shows dominant ways that Hg escapes into the environment. Note that energystar.gov gives the estimate of 103 t/yr flow of mercury into the environment. This table adds to 103.25. The importance of the source to the Hg accumulation is indicated by the % value.
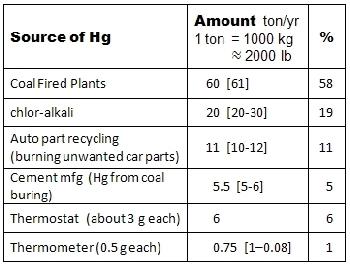 This table excludes any contribution by fluorescent lights. Let’s discuss where these numbers came from. Errors here are my own, accuracies are due to the others. This study has not been endorsed by any outside agency.
This table excludes any contribution by fluorescent lights. Let’s discuss where these numbers came from. Errors here are my own, accuracies are due to the others. This study has not been endorsed by any outside agency.
 Coal Fired Power Plants The largest source of Hg pollution is from our coal-fired plants, which means most of them. They generate 61 t/yr. 60 t/yr.
Coal Fired Power Plants The largest source of Hg pollution is from our coal-fired plants, which means most of them. They generate 61 t/yr. 60 t/yr.
Although 58% of the entire pollution pool, the industry strongly fights against any changes from the cheap way of making power because the upgrades to filter Hg out of the exhaust plume are very expensive, making coal plants about the same as nuclear plants in installed costs. The most amusing defense against exhaust filtering is the results would be solid or liquid Hg, and then we would still have to do something about that.
Chlor-Alkali This is from making chlorine products. These plants keep about 200 t in the processing inventory and typically “lose” about 10%, 20-30 t/yr. 20 t/yr.
Automotive Part Recycling Mercury is released as the unwanted car parts are burned; this accounts for 10-12 t of Hg/yr into atmosphere. 11 t/yr.
Cement manufacturing This is a problem because they burn coal to heat the kilns that process limestone. The published estimate is about 11,200 lbs, 5.6 t Hg released each year. 5.5 t/yr.
Thermostats The old style thermostats contain about 3 grams of Hg each. These are being replaced by newer technology, and in 1997, Honeywell and two other companies formed TRC Inc. to accept and recycle old thermostats of any brand. By 2010, this had grown to a consortium of 30 manufactures; by 2005 had collected 2.5 t of Hg, by 2010 this had reached 4.7 t. Both numbers represent collection rate of about 0.35 t/yr.
TRC estimates that it collects about 6% of the thermostats that are being replaced: (0.35t/yr)/0.06 = 5.8 t/yr must be sent to landfills everywhere in the U.S.. Round to 6 t/yr
A different source says that in 2002 the estimate from thermostats to landfill was 15 t/yr. If TRC was in error and its rate of voluntary recycling were 2.5%, the two estimates would be in agreement.
 Thermometers This is not much of a be source of Hg, compared to the unregulated power industry. But these things have been removed from sale all over the world. Nowadays, everyone knows that, although it causes 1% of the total mercury pollution, mercury thermometers are really, really, really bad; all our children are threatened.
Thermometers This is not much of a be source of Hg, compared to the unregulated power industry. But these things have been removed from sale all over the world. Nowadays, everyone knows that, although it causes 1% of the total mercury pollution, mercury thermometers are really, really, really bad; all our children are threatened.
A home thermometer (the type usually in the local bans that have been passed) contain about 1/2 gram of Hg.
I measured the bulb volume of my thermometer; using the low temperature density of Hg, the total quantity was very close to 0.5 g.
It is a challenge to find the associated waste volume. There are literally millions of places to find information on how to recycle and why they are naughty to own. I have had to generate my own estimates for the flow of Hg into the environment; watoxics.org gives stating data.
In 2010, poison centers about the country logged in about 15,000 calls about mercury. Assume 1% of the breakage occurrences resulted in a call. This means about 1,500,000 thermometers broke in 2010, or 750,000 g, 750 kg, 0.75 t Hg from thermometers. If the centers got 10% of the calls (extreme estimate), the lost Hg would be 0.075 t.
Check 2: a thermometer holds about 1/6 the mercury in a thermostat. My estimate is that a household would have one furnace thermostat and one thermometer. If we had 6 t/year lost from thermostats to landfill, then we should see perhaps 1 t/yr from thermometers. At this point, I would say that we are not looking at the total inventory of thermometers broken, so we should expect less than 1 t of Hg per year into the environment.
Check 3: There are about 310 M people in the U.S., and about 113 M households. Assume one mercury thermometer for every 2 households (some none, some more than 1). So the inventory is ( g=grams, hh= households, thm= thermometers)
(113 M hh) (0.5 thm/hh) (0.5 g/thm) = 28 ¼ M g = 28.25 t of Hg in hh inventory.
Assume each household breaks and buys one every 20 to 30 years. 1 – 0.9 M grams Hg disposed of each year. This is 0.9 to 1 t of Hg from thermometer disposal. ♦ As a physicist, I am happy that is agrees with the other numbers within a factor of 2 (ball park estimates being what they are). 0.75 t/yr is a realistic estimate for thermometer waste.
This is a very approximate estimate, but it is probably close to the true waste value. So 0.55 t to 1 t is probably the right interval (0.75 is nearly the geometric mean of 1 and .56).
Fluorescent lighting
Ten years ago, tube fluorescents had 20 mg of Hg, they now have about 12. At the turn of this century, CFLs had 6–10 mg, most now contain 3 mg. Note that the cheapest lights probably have more Hg than these because reliable operation is easier.
First: how many lamps does an “average” household have? One answer is about 20. Another says 76. Personally I would believe maybe 30 normal lamps. The geometric mean is about 38. Let’s use 38.
Steady state: When all the households have changed out their 38 bulbs the sales volume will be for basic replacement, and the number purchased should be approximately the number discarded. So we need to find the proprietary number of bulbs sold.
First approximation at number bulbs sold. (38 bulbs/house) (113 M households) = 4320 M. Assume half are changed out each year, expect number of around to 2 Billion bulbs sold each year, give or take some.
Best data I have found is at realneo.us in a comment by Kathryn Kranhold in 2007-Dec27 (columnist/blogger for the Wall Street Journal) titled His Bright Idea: Dominate Energy-Saving Light Bulbs. She said that the 2006 sales volume for incandescent lights was 1.5 billion with 200 M CFLs. Total is 1.7 B “luminaries” sold. In 2010, the estimate was that 25% (1 in 4) of the sales were CFLs. The true current sales numbers? Probably somewhere between 1.7 and 2.0 ×109 units.
Estimate discards when the ILBs have phased out and everyone is buying fluorescent designs (equilibrium in 2015?), I will guess the volume sold is 1 B units. Think this is twice too high? You can divide the final results by 2. Be careful with your correction factor, though. If you divide the results (see below) by 1000, only 1 M bulbs would be sold and the manufacturing would dry up from loss of sales. (model this – drop sales volume and raise prices to match … Indoor lighting becomes a rich-mans toy. I don’t even want think about that!)
We will be disposing of 1 B bulbs each year. How many will actually be recycled? If you say 100% you are in danger of utopian dreaming. I use my home base, Ann Arbor, as an example. The local drop-off center is a good drive from the center of the town. It charges $3.00 entrance fee and $1 for each fluorescent luminary received. Do not think 100% of Ann Arborites recycle. Probably only 1–10% of the discarded bulbs will be recycled and 10% is a lot higher than the recycle figure for thermostats with their 1000× mercury content. Ratios: Recycled/discarded: 10%/90%, 1%/99%.
(1×109 discards) (0.9 fraction dumped) (.003 grams each bulb) =2.7 M g of Hg
(1×109 discards) (0.99 fraction dumped) (.003 grams each bulb) =3 M g of Hg
This is 2.7 ton/yr Hg from fluorescent luminaries entering the waste stream of the United States.
Discussion Surely something is wrong with the estimate! This would mean CFL pose at least twice the hazard as mercury thermometers. To get this down to the same ¾ t Hg/yr as thermometers, we need to divide the CFL waste by 3 or 4.
You almost get there by insisting on 50% recycling (divide by 2) but this has never before been met by any other commodity. And I would insist that the 3 mg Hg is too small, considering that fluorescent tubes used in businesses and manufacturing now contain about 12 mg each. Maybe the geometric mean is the average we want … 6 mg per discard. If so, then we return to 2.7 t. Perhaps bulb sales are not currently 2 billion a year? But if not, how could Wall Mart (a single retail seller) sell 100 M bulbs in a year?
I don’t think we can estimate better than 2 to 3 tons/yr of Hg pouring into the environment without someone doing a detailed study. The results will probably be within a factor of 2 to this estimate, though. The incandescent ban will add 2% to 3% to the current mercury flow into the waste stream. Yes, worse than thermometers.
____________________________________________________
Rare-Earth Waste
 This is not too controversial. The inside layer of a tube is coated with the phosphor layer. This generates the visible light. It also contains may rare Earth heavy metals
This is not too controversial. The inside layer of a tube is coated with the phosphor layer. This generates the visible light. It also contains may rare Earth heavy metals
An example of the kinds of materials is available from Osram Sylvania. This table lists some of the most common elements. I cannot carry this much further, because there is no listing of the chemical listing by weight for each of the phosphors used in common lighting tubes. I have not seen any discussion of the effects of phosphor dispersal but there must be some. Break a tube and you find white powder dusted about. This is the powder and discarded tubes must have each element leached into the groundwater.
The Chinese experience demonstrates that elements cause problems when dispersed at high enough concentrations. Will we see water sources poisoned because of this? We have to expect 900 M tubes discarded each year (same estimate as above). Only actual testing would determine if contamination by rare earth elements will become an issue in the next 10 years. I suspect that the actual weight of the phosphors (proportional to thickness of the layer) may be corporate secrets; too heavy and visible light is absorbed, too light will cause a sunburn, if not actual eye damage (Fluorescents-5).
____________________________________________________
Summary
 By my “back of the envelop” estimate, disposal of CFL tubes poses at least twice the mercury threat as thermometers. So why would we ban thermometers because they are so bad and push fluorescents because they are so good? Neither should be our priorities: Why not drop these 1 to 3% problems and go after the big problem worth 58%, the power generation industry and its unwillingness to upgrade?
By my “back of the envelop” estimate, disposal of CFL tubes poses at least twice the mercury threat as thermometers. So why would we ban thermometers because they are so bad and push fluorescents because they are so good? Neither should be our priorities: Why not drop these 1 to 3% problems and go after the big problem worth 58%, the power generation industry and its unwillingness to upgrade?
The usual answer is because fluorescents save money. Read Fluorescents-2. If you leave CFLs on all day but switch incandescents off when not needed, CFLs cost more. Keep both on all the time, never turn off any lamp, fluorescents are the spectacular winner. If you turn them both off when not needed, you destroy the CFL long life spans, and lose.
An ideologue’s comment might be that we all just ought to do our recycling. This is the the sign of a utopian dreamer. Many social activities have been proposed that would surely lead to a wonderful existence, if only we would all just __ . Actually, people in a community act in their best personal interests; utopian dreams do not work.
We brought up a second environmental issue that I cannot estimate. The phosphors that generate the visible light are loaded with rare earths, and are dangerous in the waste stream. What will the dumping of all this new load of bulbs do to us?
Next post on fluorescents will summarize this long series, try to make a coherent pattern emerge.
Charles J. Armentrout, Ann Arbor
2011 Aug 16
This is listed under Technology …thread Technology > Fluorescents
Have a comment? Click on the title of this post, go to bottom, let us know.
Related posts: Click the INDEXbutton under the Banner picture

